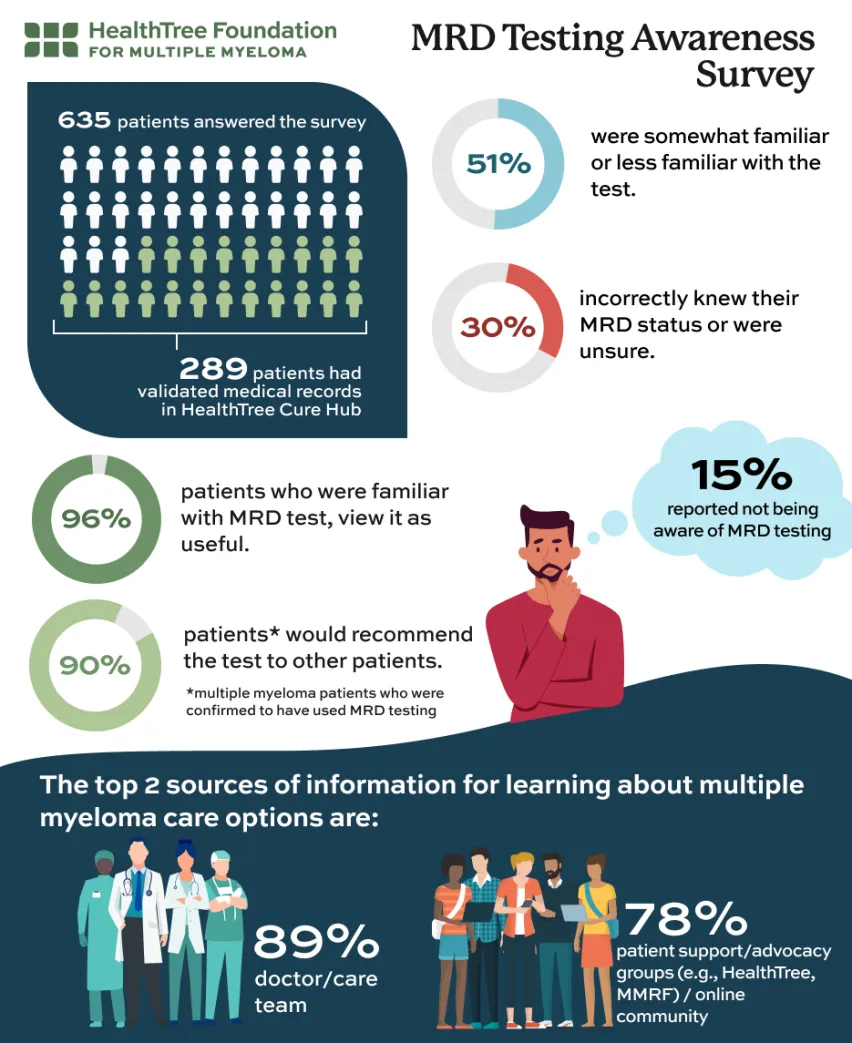
▲A U.S. survey of 635 multiple myeloma patients revealed that half had limited knowledge of MRD testing, yet most who underwent it reported both clinical and emotional benefits.
Source:What Multiple Myeloma Patients Think About MRD Testing
Multiple myeloma (MM), the second most common blood cancer globally, remains complex and incurable despite significant therapeutic advances. Recent developments in minimal residual disease (MRD) testing have revolutionized disease monitoring and treatment strategies. This article explores MRD’s pivotal role in managing MM progression.

▲Dr. Brian GM Durie, a myeloma expert, highlights in a patient Q&A video that mass spectrometry (MS) is emerging as a powerful tool for tracking ultra-low myeloma levels as the technology becomes more accessible.
Source: Should patients use mass spectrometry to measure myeloma protein levels?
What Is MRD?
MRD refers to residual cancer cells persisting after treatment. MRD testing detects as few as 1 myeloma cell in 1 million healthy cells—far surpassing traditional methods like bone marrow biopsies. Even minimal residual cells can trigger relapse, making high-sensitivity MRD testing vital for evaluating treatment efficacy, predicting relapse risk, and guiding clinical decisions.
Applications of MRD Testing in Multiple Myeloma
-
Treatment Response Assessment
-
MRD negativity (no detectable cancer cells) correlates with better prognosis, while MRD positivity indicates higher relapse risk.
-
Clinicians use MRD status to refine treatment plans, such as adjusting therapy intensity or duration.
-
-
Relapse Risk Prediction
-
MRD-negative patients show longer progression-free survival (PFS) and overall survival (OS) compared to MRD-positive patients.
-
-
Guiding Treatment Decisions
-
MRD-negative patients may reduce treatment intensity to minimize toxicity, while MRD-positive patients may benefit from intensified therapies like CAR-T or novel targeted drugs.
-
-
Drug Development
-
MRD negativity is increasingly used as a clinical trial endpoint to evaluate new therapies, accelerating drug approvals.
-
【FAQs about MRD】
Is MRD testing widely used in clinical practice?
Currently, MRD testing is primarily utilized in clinical trials to monitor remission status and relapse likelihood. Its integration into routine care is pending standardization.
Can MRD serve as an early endpoint in clinical trials?
In April 2024, the FDA’s Oncologic Drugs Advisory Committee unanimously endorsed MRD as an early endpoint for accelerated drug approvals in myeloma trials. This shift allows researchers to predict long-term outcomes like PFS and OS more efficiently.
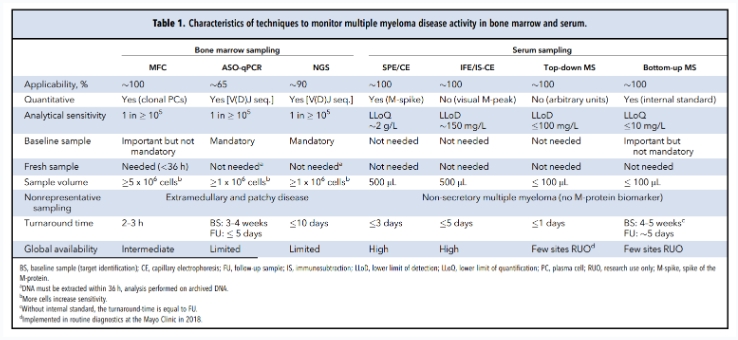
Current MRD Detection Methods
Who Should Consider MRD Testing?
-
Post-treatment patients evaluating response.
-
High-risk patients requiring relapse monitoring.
-
Participants in clinical trials assessing drug efficacy.
Key Trends in Multiple Myeloma
-
Ethnic Disparities: African Americans face twice the incidence rate compared to Caucasians.
-
Survival Improvements: Since 2000, advancements in therapies have turned MM into a more manageable chronic condition.
-
Early Detection: 20-30% of patients are high-risk, emphasizing the need for timely intervention.


MarkerX: Your Partner in the Fight Against Multiple Myeloma
MarkerX’s MM-MRD test:
- Accurately assess your risk of multiple myeloma relapse
- Is a simple blood test with fast results
- Helps your doctor personalize your treatment
Contact us to learn more.
Reference
- MRD and Mass Spectrometry Testing,International Myeloma Foundation Headquarters。
- What Is MRD Testing?,HealthTree Foundation,May 28, 2024
- Thank to Myeloma Action Month‘s information and social media card design.

